You’ll have 1,900+ minutes to spend at ESGCT.
And we have a great suggestion for 20 of them.
Booth B10 & B12 | Sevilla, Spain | October 7-11, 2025
You should expect a lot of the manufacturing partner you entrust with your AAV gene therapy. We’re up to the task. And we’d welcome a few minutes of your time at ESGCT to explain why.
Forecast your serotype’s performance from our 1,500+ AAV runs
Leverage our purpose-built AAV platform designed for best-in-class speed to scale
Partner with our team of experts that has 100% success rate in IND/IMPD dossier submissions
Schedule A Meeting

Presented by César Trigueros, PhD
Chief Scientific Officer, Viralgen
SESSION 6a
A Strategic Platform Roadmap for Commercializing Gene Therapy
Wednesday, October 8th, 18:00-19:30 · PARALLEL




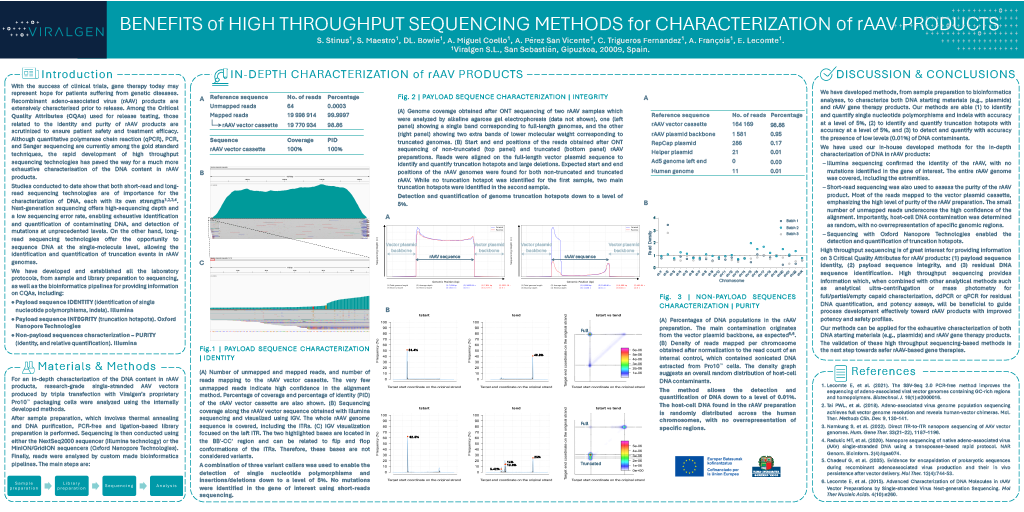
Benefits of High-Throughput Sequencing-Based Methods for In-Depth Characterization of Recombinant AAV Products
Presented by Sonia Stinus, PhD.
Poster # AMA905
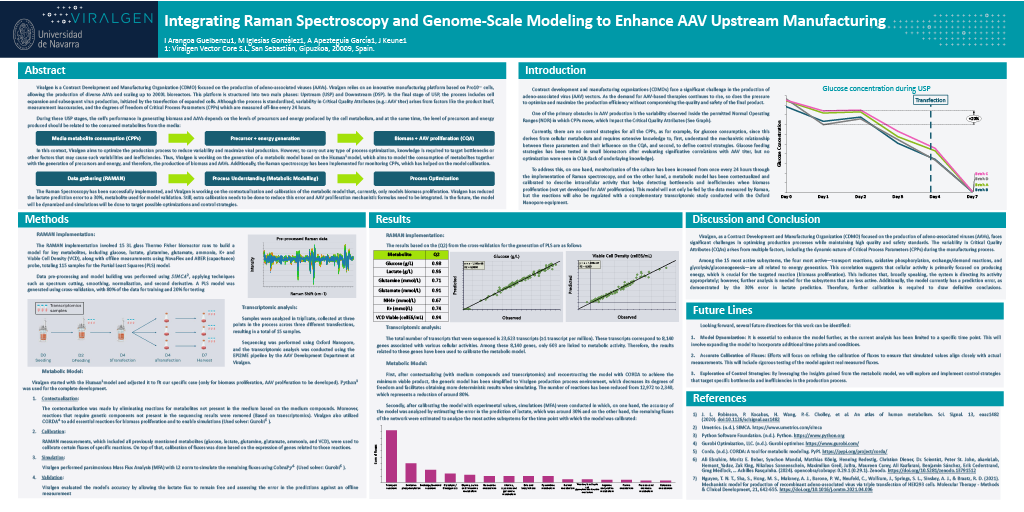
Integrating Raman Spectroscopy and Genome-Scale Modeling to Enhance AAV Upstream Manufacturing
Presented by Itsasne Arangoa.
Poster # AMA1539
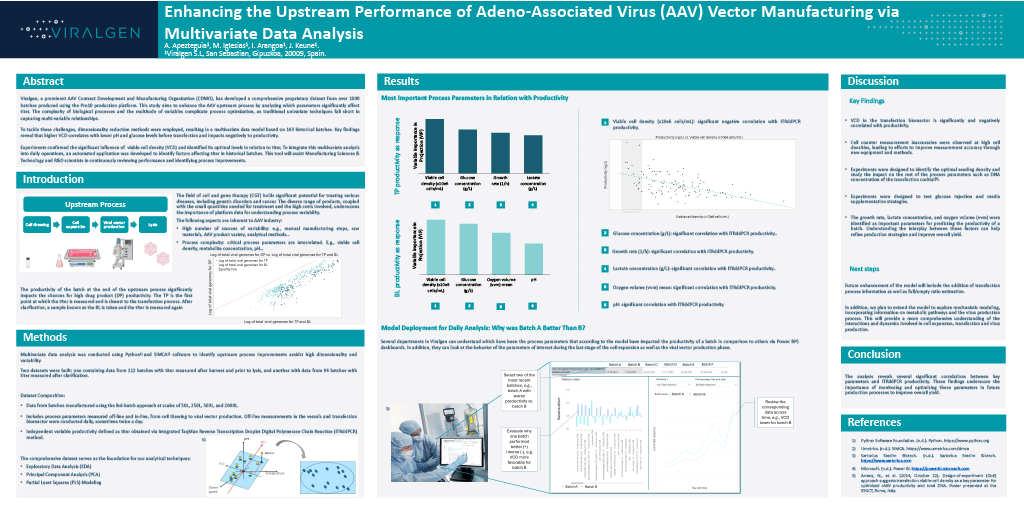
Enhancing the Upstream Performance of Adeno-Associated Virus (AAV) Vector Manufacturing via Multivariate Data Analysis
Presented by Ainara Apezteguia.
Poster # AMA1624

Leveraging Historical Data and Percentile based Modeling for Improved AAV Production Scaling
Presented by Itsasne Arangoa.
Poster # AMA1546
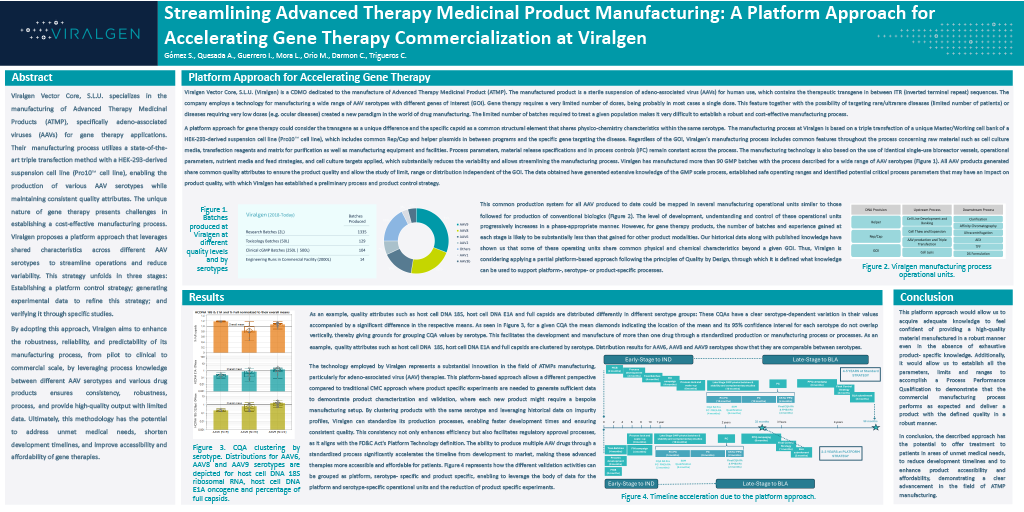
Streamlining Advanced Therapy Medicinal Product Manufacturing: A Platform Approach for Accelerating Gene Therapy Commercialization at Viralgen
Presented by Cesar Trigueros, PhD.
Poster # AMA988





